Nineteenth century geologists recognized that rocks formed slowly as mountains eroded and sediments settled on the ocean floor. But they could not say just how long such processes had taken, and thus how old their fossils were. Darwin had argued that the Earth was immensely old — which gave his gradual process of evolution plenty of time to unfold. The great physicist Lord Kelvin had countered that the planet was actually relatively young — perhaps 20 million years old. He came up with that figure by estimating how long it had taken for the planet to cool down to its current temperature from its molten infancy. But Kelvin didn’t, and couldn’t, know that radioactive atoms such as uranium were breaking down and keeping the planet warmer than it would be otherwise.
Nineteenth century geologists recognized that rocks formed slowly as mountains eroded and sediments settled on the ocean floor. But they could not say just how long such processes had taken, and thus how old their fossils were. Darwin had argued that the Earth was immensely old — which gave his gradual process of evolution plenty of time to unfold. The great physicist Lord Kelvin had countered that the planet was actually relatively young — perhaps 20 million years old. He came up with that figure by estimating how long it had taken for the planet to cool down to its current temperature from its molten infancy. But Kelvin didn’t, and couldn’t, know that radioactive atoms such as uranium were breaking down and keeping the planet warmer than it would be otherwise.
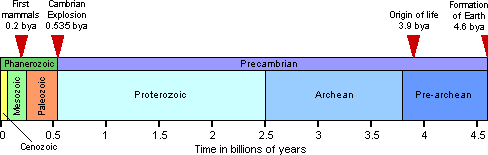
An older Earth
At the dawn of the twentieth century, physicists made a revolutionary discovery: elements are not eternal. Atoms can fuse together to create new elements; they can also spontaneously break down, firing off subatomic particles and switching from one element to another in the process (see figure, right). While some physicists used these discoveries for applications ranging from nuclear weapons to nuclear medicine, others applied them to understanding the natural world. The sun was once thought to burn like a coal fire, but physicists showed that it actually generates energy by slamming atoms together and creating new elements. The primordial cloud of dust that came to form the Earth contained unstable atoms, known as radioactive isotopes. Since its birth, these isotopes have been breaking down and releasing energy that adds heat to the planet’s interior.
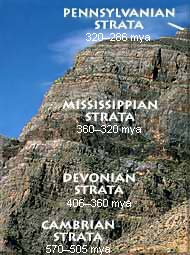
Radioactivity also gave the history of life an absolute calendar. By measuring the atoms produced by these breakdowns inside rocks, physicists were able to estimate their ages (right). And by comparing the ratios of those atoms to atoms from meteorites, they could estimate how long ago it was that the Earth formed along with the rest of the solar system. In 1956 the American geologist Clair Patterson (left) announced that the Earth was 4.5 billion years old. Darwin had finally gotten the luxury of time he had craved.
Ancient life
The dates that radioactive clocks have put on evolutionary history are astonishing. Life is well over 3.5 billion years old, and until about 600 million years ago, the planet was dominated by microbes. Radioactive clocks have shown that evolution can change its pace — the Cambrian Explosion of about 535 million years ago saw the relatively rapid emergence of many major lineages of animals in just a few million years. Mammals, which for 150 million years had been small, rodent-sized creatures, rapidly evolved to massive proportions in the wake of the Cretaceous-Tertiary extinction 65 million years ago. Geological timekeeping continues to be a lively science, with new methods emerging all the time. Some of these methods have helped to pin down the evolution of our hominid ancestors; anatomically modern humans evolved about 100,000 years ago. While that’s nearly 20 times older than the Earth was once thought to be, it’s a geological eye blink.