Diseases that pose global health threats — like HIV, malaria, and tuberculosis — regularly make the news. Last month, for example, saw reports that HIV infection rates in the US are up, that malaria statistics worldwide are down, and that the distribution of medicines to treat the three diseases had improved. Diseases with such epidemic proportions tend to make us focus on the near future: Regardless of how we wound up in this situation, what can we do now to prevent future infections and deaths? But sometimes — especially in the case of populations coevolving with disease pathogens — a glimpse of our evolutionary past, can be surprisingly informative.
Where's the evolution?
An international group of researchers recently revealed that HIV’s deadly assault on sub-Saharan Africa (which now accounts for almost 70% of HIV infections worldwide) may have been accelerated by that population’s past evolution. In that region, nearly the entire population carries a mutation to a gene known as DARC. The mutation — a single letter change of the gene’s sequence — is, however, extremely rare in people descended from ancient European, Asian, and American populations. Why the genetic dividing line? It could just be a quirk of evolution — after all, plenty of genetic differences between populations are simply the neutral result of historical accidents — but in this case, the difference seems to be more significant.
The mutation occurs in a region of the genome that controls production of the DARC protein and prevents that protein from appearing where it usually would — on the surface of red blood cells. Because the malaria species Plasmodium vivax normally uses DARC to enter red blood cells, people who carry this mutation are resistant to one strain of malaria. This particular strain of malaria is not a major killer in Africa today, but was likely more threatening in the past. Biologists reason that the DARC mutant arose in Africa through random mutation and began to spread through the process of natural selection. Individuals carrying the mutation — especially those carrying two copies of it — were better able to survive, reproduce, and pass the genetic quirk on to their offspring. Like the more familiar sickle cell mutation, which also confers malaria resistance, the mutant DARC gene is most common in Africa because of selection caused by the malaria parasite.
Unfortunately, the results of that selection now seem to be contributing to the devastation of African populations by a more modern killer. New research has highlighted a side effect of the DARC mutation that evolution — having no foresight — could not have anticipated: added susceptibility to HIV infection. The nature of the interplay between DARC and the HIV virus is not yet clear, but the results of this interaction are potentially alarming. Researchers studied a group of African American men in the US Air Force, some of whom carried the DARC mutation and some of whom did not. Men with the mutation were 40% more likely to have acquired HIV. If the DARC mutation is indeed behind this added vulnerability, it would account for 11% of the HIV infections in Africa! The researchers also discovered a somewhat paradoxical effect associated with the mutation. Men with the mutation survived HIV longer than infected men lacking the mutation.
When the CCR5-delta32 mutation was first discovered — and when its restriction to European populations was identified — scientists hypothesized that the mutation had been favored because it offered resistance to an intense epidemic largely restricted to Europe: the bubonic plague pandemic of the Middle Ages. However, when scientists studied ancient DNA from 2900-year-old skeletal remains, they found that the mutation was common in European populations long before the plague epidemics. Further research and several lines of evidence suggested that smallpox may have been the more likely culprit. New research has offered yet another hypothesis. The genetic variation associated with the mutation is similar to the genetic variation associated with neutral mutants. Perhaps, the CCR5 mutant drifted to high frequencies in Europe through nothing more than luck. Researchers continue to investigate the possibilities — but meanwhile, there is little doubt that the CCR5 mutant offers a significant survival advantage to those at risk of HIV infection.
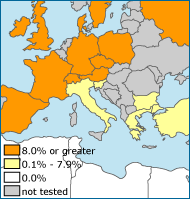
Obviously, the most important factor in determining whether or not an individual contracts HIV is contact with the virus through bodily fluids, but as research on the disease has progressed, scientists have learned that many other factors, including genetic ones, affect an individual’s chance of contracting the disease after exposure. These genetic factors may be linked to the evolutionary history of populations. For example, the CCR5-delta32 mutation, which provides almost complete protection from HIV when two copies are carried, seems to have originated in a European population more than 5000 years ago. Since then, the variant has risen to relatively high frequency in Europe (5-14%), through either genetic drift or selection, and is now found almost exclusively in individuals descended from European populations. The DARC mutation provides another, more foreboding, example of the role that evolutionary history can play in the course of modern diseases. The traces of past selection on African populations seem to be intensifying their struggle with the HIV epidemic today.
Knowledge of how the genetic variants resulting from our past evolution influence the spread and progression of modern diseases can inform approaches we take to treating and preventing them. Genetic variants like the DARC and CCR5 mutations could affect how individuals bearing them respond to HIV treatment and whether or not a vaccine is effective for them. New research needs to account for this possibility. Furthermore, understanding what makes some individuals more or less susceptible to a particular disease can inspire the design of new treatments or vaccines. Evolutionary history is critical not just for an understanding of where human populations have been, but for charting a healthy course into the future.
Primary literature:
- Carter, R., and Mendis, K. N. (2002). Evolutionary and historical aspects of the burden of malaria. Clinical Microbiology Reviews 15(4):564-594. Read it »
- He, W., Neil, S., Kulkarni, H., Wright, E., Agan, B.K., Marconi, V.C., Dolan, M.J., Weiss, R.A., and Ahuja, S.K. (2008). Duffy antigen receptor for chemokines mediates trans-infection of HIV-1 from red blood cells to target cells and affects HIV-AIDS susceptibility. Cell Host & Microbe 4(1):52-62. Read it »
- Hummel, S., Schmidt, D., Kremeyer, B., Herrmann, B., and Oppermann, M. (2005). Detection of the CCR5-delta32 HIV resistance gene in Bronze Age skeletons. Genes and Immunity 6(4):371-374. Read it »
- Sabeti, P. C., Walsh, E., Schaffner, S. F., Varilly, P., Fry, B., Hutcheson, H. B., Cullen, M., Mikkelson, T. S., Roy, J., Patterson, N. Cooper, R., Reich, D., Altshuler, D., O'Brien, S., and Lander, E. S. (2005). The case for selection at CCR5-delta32. PLoS Biology 3(11):1963-1969. Read it »
News articles:
- A description of new research on the DARC mutation from Science News
- A summary of the discovery from the BBC
Understanding Evolution resources:
- Review a description of how the sickle cell mutation winds up affecting the phenotype of an individual. Write a similar description of what we know about how the DARC mutation affects an individual’s phenotype. Be sure to begin at the level of the DNA.
- The DARC mutation described here is more common in African populations because of the past action of natural selection. Research and describe another trait that is more common in some populations than others because of past episodes of natural selection.
- Review some background information on natural selection. Explain how the DARC mutation would spread through a population severely affected by Plasmodium vivax but not affected by HIV. Make sure to include the concepts of variation, selection, and inheritance in your explanation.
- Imagine that the population described in the question above lives in modern day Canada, where malaria is not a problem but HIV is a potential threat. How would that change your answer to the question above?
- Review the boxed text above, which describes scientists’ ideas about why the CCR5 mutation is found at higher frequencies in European populations than in the rest of the world. Scientists discovered a relatively high frequency of the mutation in 2900-year-old skeletal remains. Explain why this evidence argues against the idea that the mutation rose to high frequencies because it offered protection from the plague.
- Teach about human evolution at the molecular level: In this lesson for grades 9-12, students investigate variation in the beta globin gene by identifying base changes that do and do not alter function, and by using several internet-based resources to consider the significance in different environments of the base change associated with sickle cell disease.
- Teach about another example of human evolution at the molecular level: This article for grades 9-12 explains that the ability to digest milk is a recent evolutionary innovation that has spread through some human populations — much as the DARC mutation spread through African populations.
- Carter, R., and Mendis, K. N. (2002). Evolutionary and historical aspects of the burden of malaria. Clinical Microbiology Reviews 15(4):564-594.
- Harris, G. (2008, September 11). Detailed study of spread of H.I.V. in U.S. The New York Times. Retrieved September 26, 2008 from The New York Times
- He, W., Neil, S., Kulkarni, H., Wright, E., Agan, B.K., Marconi, V.C., Dolan, M.J., Weiss, R.A., and Ahuja, S.K. (2008). Duffy antigen receptor for chemokines mediates trans-infection of HIV-1 from red blood cells to target cells and affects HIV-AIDS susceptibility. Cell Host & Microbe 4(1):52-62.
- Hummel, S., Schmidt, D., Kremeyer, B., Herrmann, B., and Oppermann, M. (2005). Detection of the CCR5-delta32 HIV resistance gene in Bronze Age skeletons. Genes and Immunity 6(4):371-374.
- MacFarquhar, N. (2008, September 4). Donors' aid to poor nations declines, U. N. reports. The New York Times. Retrieved September 26, 2008 from The New York Times
- McNeil Jr., D. G. (2008, September 22). Revisions sharply cut estimates on malaria. The New York Times. Retrieved September 26, 2008 from The New York Times
- Sabeti, P. C., Walsh, E., Schaffner, S. F., Varilly, P., Fry, B., Hutcheson, H. B., Cullen, M., Mikkelson, T. S., Roy, J., Patterson, N. Cooper, R., Reich, D., Altshuler, D., O'Brien, S., and Lander, E. S. (2005). The case for selection at CCR5-delta32. PLoS Biology 3(11):1963-1969.
- UCSF Center for HIV Information. (2008, September). Sub-Saharan Africa. HIV InSite. Retrieved September 26, 2008 from HIV InSite
